The Bill & Melinda Gates Foundation is joining Pfizer Inc. and the Children’s Investment Fund Foundation as part of its $1 billion dollar effort to make it more accessible for women in poor countries to receive contraceptives.
Depo-Provera, the injectable contraceptive has been a favorite among women in Asia and Africa. Getting these shots, however, would require a lengthy journey to a hospital or clinic where a health worker must inject the drug deep into the muscle.
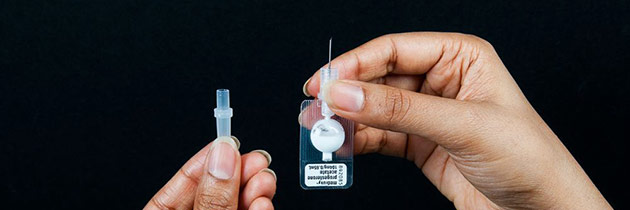
Sayana Press, however, the new incarnation of the drug comes in a syringe consisting of a needle attached to a plastic bubble. In order to administer the job the only thing that needs to be done is squeezing the plastic bubble. This process is so easy that even minimally trained workers can do it.
CEO of the Children’s Investment Fund Foundation, Michael Anderson said the real innovation of the product is that it’s so simple to use.
Production of the Sayana Press drug by Pfizer will be subsidized by the Gates Foundation and the Children’s Investment Fund Foundation. Pfizer agreed to sell the drug to groups like the U.S. Agency for International Development and governments for $1 per a dose of three-months.
A spokeswoman of the Gates Foundation said the drug will be made available to women in 69 of the poorest countries on Earth at little or no cost at all.
The wife of Microsoft founder Bill Gates, Melinda Gates made family planning one of the foundation’s highest priorities back in 2012. The philanthropy is committed on spending $1 billion in order to make contraceptives available by 2020 to 120 million women.
The global development chief of The Gates Foundation, Chris Elias said in a briefing on Thursday:
“When women have access to the means to time and space their pregnancies in a healthy way, we see improvements in maternal health, child health and in the economic prosperity of nations,”
The Seattle nonprofit organization PATH developed the syringe called Uniject.
The Gates Foundation is sponsoring numerous trials in Africa in order to see if it is feasible and practical for women to administer the drugs themselves in their own homes.
The Sayana Press contraceptive is not licensed for use in the U.S.